![Units of rate constant a Order Units of rate Constant Reaction moll-1 Zero order reaction s (moll-1)0 -1,0 = moll-Is-1 moll! -X 1 -1 First order reaction = S s (moll-1)] Second Units of rate constant a Order Units of rate Constant Reaction moll-1 Zero order reaction s (moll-1)0 -1,0 = moll-Is-1 moll! -X 1 -1 First order reaction = S s (moll-1)] Second](https://toppr-doubts-media.s3.amazonaws.com/images/4986560/04db6649-cd37-4cce-83ea-218d6083d9fd.jpg)
Units of rate constant a Order Units of rate Constant Reaction moll-1 Zero order reaction s (moll-1)0 -1,0 = moll-Is-1 moll! -X 1 -1 First order reaction = S s (moll-1)] Second
99 1 mol N2 and 3 mol H2 are placed in a closed container at a pressure of 4 atm. The pressure falls to 3 atm at the same temperature when the following equillibrium is attained

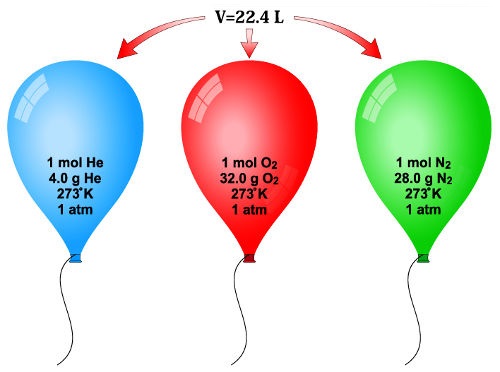
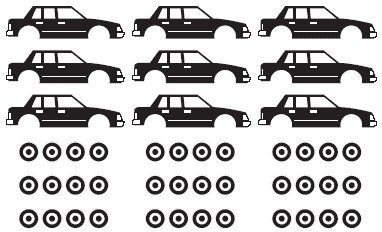
La masa molar es la masa molecular expresada en gramos 1 mol de átomos 12 C es = 6.022 x 1023 átomos = 12.00 g 1 átomo 12 ... | Masa molar, Masa molecular, Masa
![Units of rate constant a Order Units of rate Constant Reaction moll-1 Zero order reaction s (moll-1)0 -1,0 = moll-Is-1 moll! -X 1 -1 First order reaction = S s (moll-1)] Second Units of rate constant a Order Units of rate Constant Reaction moll-1 Zero order reaction s (moll-1)0 -1,0 = moll-Is-1 moll! -X 1 -1 First order reaction = S s (moll-1)] Second](https://toppr-doubts-media.s3.amazonaws.com/images/12772699/f958ec6c-f6cc-4038-9fbd-686848a6e701.jpg)




![Solved Given that Delta H degree r[H(g)] = 218.0 kJ. mol-1 | Chegg.com Solved Given that Delta H degree r[H(g)] = 218.0 kJ. mol-1 | Chegg.com](https://d2vlcm61l7u1fs.cloudfront.net/media/f73/f73ff219-6265-444d-89c1-620f5d2f2822/php7wff8s.png)













![🌐¿Qué es el Mol? Número de AVOGADRO💫 [Fácil y Rápido] | QUÍMICA | - YouTube 🌐¿Qué es el Mol? Número de AVOGADRO💫 [Fácil y Rápido] | QUÍMICA | - YouTube](https://i.ytimg.com/vi/qdTVrJzCsC0/maxresdefault.jpg)